Last updated on July 14th, 2024 at 05:57 pm
Solid State

A Solid State is one of the four fundamental states of matter which has a rigid structure and closely packed molecules.
Properties of Solid State:
Classification of Solids
| Properties | Crystalline Solid | Amorphous Solid |
|---|---|---|
| Shape | Definite geometric shape.Arrangement is ordered and repetitive in 3-D | Indefinite/Irregular shape |
| Melting Point | Sharp melting point | Don’t have a sharp M.P and softens over a range of temperature |
| Cleavage Property | When cut with a sharpened edge tool, the pieces obtained will be smooth and plain | When cut with a sharpened edge tool, the pieces obtained will have irregular surface |
| Anisotropy | Anisotropic | Isotropic |
| Heat of Fusion | They have a definite enthalpy of fusion | They don’t have a definite enthalpy of fusion |
| Nature | True Solids | Pseudo Solid |
| Order in arrangement of constituent particles | Long Range Order | Short Range Order |
| Examples | NaCl, Quartz, Naphthalene, Benzoic Acid, Copper etc | Quartz Glass, Rubber, Plastic, Teflon, Cellophane, PVC, Polymers etc |
Note:
? Quartz is a crystalline solid and Quartz glass is an amorphous solid
? The glass on old monuments appears milky because over a long range of time, due to continuous heating and cooling, it acquires some crystalline characters being an amorphous solid. This process is called Annealing
? Amorphous solids are called pseudo solids because they have a tendency to flow like liquids.
? The glass in older monuments is thicker at bottom from the top because over a long period of time, glass being an amorphous solid, which is also a pseudo solid, flows in the downward direction.
Isotropy
Isotropy is the nature of a solid which means uniformity in all directions.
Their properties such as mechanical strength, refractive index, electrical conductivity remain the same in all directions.
This is because there is no long range order in the arrangement of particles.
Amorphous Solids are isotropic in nature whereas crystalline solids are anisotropic in nature
Classification of Crystalline Solids
- Molecular Solids– Molecules are the constituent particles.
- Non-Polar Molecular Solid- Atoms and molecules are held by weak dispersion or London forces. They are soft and electric insulators. They have a low melting point. Example- H2, Cl2, I2
- Polar Molecular Solid– They are formed by polar covalent bonds and are held together by relatively stronger dipole-dipole interactions. They are soft and electric insulators. Examples- Spoiled SO2, Solid NH3 etc.
- Hydrogen Bonded Molecular Solid– Molecules have polar covalent bonds between H and N,O and F. They are electric insulators.They are soft solids under room temperature.
2. Ionic Solids – Ions are the constituent particles.
- Formed by 3-D arrangement of cations and anions
- They have strong coulombic force
- Hard and brittle in nature
- High melting and boiling points
- Electrical insulators in solid state whereas conductors in molten and aqueous state
- Example- (NH4)3PO4, LiBr etc
3. Metallic Solids – They have an orderly collection of positive ions surrounded by and held together by a sea of electrons.
- Free mobile electrons are responsible for high conductivity of metals Lustrous Malleable and ductile
4. Covalent or Network Solids – Particles are covalently bonded.
- Hard and brittleAtoms are held strongly Electrical InsulatorsExample- Diamond, Graphite, Quartz
Crystal Lattices and Unit Cell
When each constituent particle in a crystal is depicted as a point in a 3-D arrangement, the arrangement is called a crystal lattice.
14 possible 3D lattices are called Bravais Lattices and the characteristics are:
Unit Cell
It is the smallest part of a crystal lattice which, when repeated in all possible different directions, generates the entire lattice. Example- Primitive Unit Cell, Centre Unit Cell.
? Primitive Unit Cells – When constituents particles are present only and only on the corner positions of a unit cell, it’s called a primitive unit cell.

? Centred Unit Cells – When a unit cell contains constituent particles present at positions other than corners in addition to those at corners, it’s called a centred unit cell. They are of 3 types-
- Body-Centered Unit Cell (BCC):
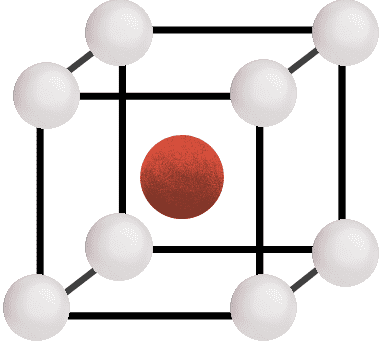
It contains one constituent particle at its body-centre besides the ones that are at its corners
2. Face-Centred Unit Cell (FCC):

It contains one constituent particle present at the center of each face, besides the ones that are at its corners
3. End-Centred Unit Cell (ECC):

One constituent particle is present at the center of any two opposite faces besides the ones present at its corners
An unit cell is characterised by:-
- It’s dimensions along the 3 edges a,b and c
- Angles between the edges, ɑ(between a and b), β(between b and c) and ℽ(between a and b)
| CRYSTAL SYSTEM | POSSIBLE VARIATION | EDGE LENGTH | AXIAL LENGTH | EXAMPLES |
|---|---|---|---|---|
| Cubic | Primitive, BCC, FCC | a = b = c | ɑ = β = ℽ = 90° | NaCl, ZnS, Cu |
| Tetragonal | Primitive, BCC | a = b ≠ c | ɑ = β = ℽ = 90° | White Tin, SnO2 |
| Orthorhombic | Primitive, BCC, FCC, ECC | a ≠ b ≠ c | ɑ = β = ℽ = 90° | Rhombic Sulphur, KNO3 |
| Hexagonal | Primitive | a = b ≠ c | ɑ = β = 90°ℽ = 120° | Graphite, ZnO |
| Trigonal or Rhombohedral | Primitive | a = b = c | ɑ = β = ℽ ≠ 90° | Calcite, Cinnabar |
| Monoclinic | Primitive, ECC | a ≠ b ≠ c | ɑ = ℽ = 90°β ≠ 90° | Monoclinic Sulphur |
| Triclinic | Primitive | a ≠ b ≠ c | ɑ ≠ β ≠ ℽ ≠ 90° | Potassium Dichromate, H3BO3 |
Number Of Atoms In A Unit Cell
Primitive Unit Cell
- It has atoms only at its corner.
- Each atom is shared between 8 adjacent unit cells
- Only ⅛th of an atom actually belongs to a particular unit cell
- There are 8 atoms on the corner of a cubic unit cell
- Total number of atom in one unit cell – 8x⅛ = 1 atom
Body Centered Cubic Unit Cell
- It has an atom at each corner and also one at the body centre
- Total number of atom in one unit cell
- Corners – 8x⅛ = 1 atom
- Body Centre – 1×1 = 1 atom
- Total – 1 + 1 = 2 atoms
Face-Centered Cubic Unit Cell
- Contains atoms at the corners and at the centre of all faces
- Each atom at the face is shared between two unit cell
- Only ½th of each atom belongs to a unit cell
- Total number of atom in one unit cell
- Corners – 8x⅛ = 1 atom
- Face-Centres – 6x½ = 3 atoms
- Total – 1 + 3 = 4 atoms
Coordination Number
- The number of nearest neighbours around a particle is called coordination number
- Coordination number in :-
- 1D – 2
- 2D – a. Square Close Packing – 4 b. Hexagonal Close Packing – 6
Closed Packed Structures
- Closed Packing in 1D
- Closed Packing in 2D
- Square Close Packing – AAAA Type
- Hexagonal Close Packing – ABAB Type
- Closed Packing in 3D
- Square Close-Packed Layers – AAAA Type
- Hexagonal Close-Packed Layers – ABAB Type

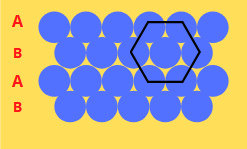
Voids
The vacant spaces between the constituent particles in a close packed structure are called voids.
There are two types of voids –
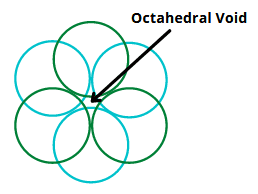

Formula Of A Compound And Number Of Voids Filled
- Number of octahedral voids present in a lattice is equal to the number of close packed particles
- Number of tetrahedral voids generated is twice this number
Packing Efficiency
The total percentage of space filled by the particle is called Packing Efficiency
Packing Efficiency in :-
✅ HCP and CCP Structures
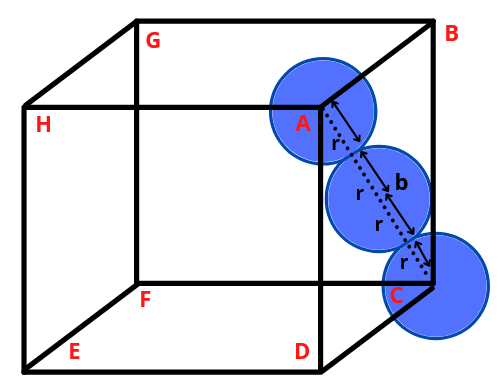
- Let the edge length of unit cell be ‘a’
- In ΔABC, AC2 = b2 = BC2 + AB2 = a2 + a2 = 2a2
- b = √2a
- If ‘r’ is the radius of sphere, b = 4r =√2a
- a = 2√2r
- Void Percentage = 100 – 74 = 26%
✅ Body-Centred Cubic Structure
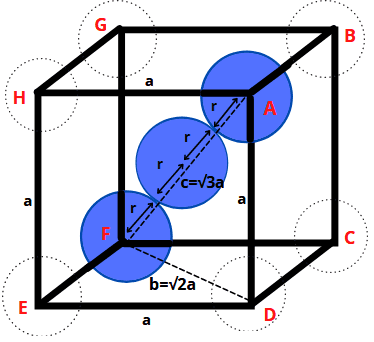
- In ΔEFD, b2 = a2 + a2 = 2a2 b = √2a
- In ΔAFD, c2 = a2 + b2 = a2 + 2a2 = 3a2
- c = √3a
- Length of body diagonal c is equal to 4r
- √3a = 4r
-
- Void Percentage = 100 – 68 = 32%
✅ Simple Cubic

- a = 2r
- Packing Efficiency = Volume of one atom Volume of cubic unit cellx 100%
- Void Percentage = 100 – 52.4 = 47.6%
☸ Calculating Density
- Edge length = a
- Volume of unit cell = a3
- Mass of unit cell = Number of atoms in unit cell x Mass of each atom = Z x m
- m = M/NA
- Density = Mass of unit cell / Volume of unit cell

☸ Imperfection in solid
- Stoichiometric Defect – They don’t disturb the stoichiometry of the solid. Also called thermodynamic or intrinsic defect
Types of Stoichiometric
⛔ Vacancy Defect –

- When any lattice site is vacant, the crystal is said to have a vacancy defect.
- It results in a decrease in density.
- Developed due to heating of solid.
⛔ Interstitial Defect –

- When a particle occupies an interstitial site, the crystal is said to have an interstitial defect.
- This increases the density of the solid.
⛔ Frenkel Defect –

- Smaller ion (cation) is dislocated from its position to an interstitial site
- This creates a vacancy defect at its original site and an interstitial defect in its new location
- Also called dislocation defect
- No change in the density
- Shown by ionic substances having large variation in the cationic and anionic size
- Example – ZnS, AgCl, AgBr etc
⛔ Schottky Defect –

- The number of missing cations and anions are same
- Electrical neutrality is maintained
- Density gets decreased
- Shown by ionic substance in which the size of cations and anions are identical
- Example – NaCl, AgBr, KCl etc
2. Impurity Defect –
- Example – Sr2+ occupies the 2 Na+ sites when SrCl2 is mixed in a small amount with molten NaCl
3. Non-Stoichiometric Defect – Defects which affect the stoichiometry of a solid
Non-Stoichiometric defects types :-
? Metal Excess Defect due to anionic vacancies

- When NaCl crystals are heated in Sodium vapour, sodium atoms are deposited on the surface of the crystals
- Cl– ions diffuse to the surface to combine with the sodium atoms and form NaCl
- Na loses one electron which diffuses and reaches the vacant site created by Cl
- This site created by the electron is called the F-centre (Farbenzenter centres)
- These sites results in the colour of a crystal due to the excitation of electrons when they absorb energy from light
Example – NaCl shows yellow colour, LiCl shows pink colour, KCl shows violet colour etc.
? Metal Excess Defect due to extra cations at interstitial sites
- Zinc Oxide is white in colour at room temperature
- On heating it loses oxygen and turns yellow
- There is excess of Zn in the crystal and formula becomes Zn1+xO
- Excess Zn2+ moves to interstitial sites
- ZnO Zn2+ + 12O2 + 2e–
? Metal Deficiency Defect
- The amount of metal present in the crystal is less then the actual stoichiometric ratio
- Example – Fe2+ cations are replaced by Fe3+. This results in the configuration of Fe0.93O-Fe0.96O rather than FeO
Note:
– AgBr can show both Frenkel and Schottky Defect
– Frenkel defect is not shown by pure alkali metal halide because the alkali metal ions have large size which cannot fit into the interstitial sites
Electrical Property
Solids are classified into 3 types based on their electrical conductivity:-
- Conductors – Conductivity ranges between 104-107 ohm-1m-1
- Insulators – Conductivity ranges between 10-20-10-10 ohm-1m-1
- Semiconductors – Conductivity ranges between 10-6-104 ohm-1m-1
- Conductivity of Metals
- Metals conduct electricity in molten as well as solid state
- Atomic orbitals of metal forms molecular orbitals which are close to each other forming bands
- If this band is partially or fully overlaps the conduction band, then electrons can flows easily after an electric field is applied
2. Conduction in Insulators
- If the gap between the band and the conduction band (Forbidden Energy Gap) is large, then the electrons won’t be able to gain enough energy by any means to reach the conduction band.
- Therefore they don’t conduct any electricity
3. Conductivity of Semiconductors
- The Forbidden Energy Gap between the conduction and the valence band is small
- Some electrons might get enough energy to reach the conduction band and some might not reach it
- Therefore their conducting property is between a conductor and an insulator
- Materials showing this property are called intrinsic semiconductors. Example – SIlicon and Germanium
- Intrinsic semiconductors have very low conductivity and hence are used very less in day to day appliances
- To increase the conductivity of intrinsic semiconductors, these are doped with impurities of group 13 and 15 elements
- Doping is the deliberate addition of impurities (Group 13 or 15 elements) in an intrinsic semiconductor to increase its conductivity
- Semiconductors are of 2 types based on the doping:-
➡️ N-Type Semiconductor
- Intrinsic semiconductors when doped with gr-15 elements (P, As, Sb), form n-type semiconductor
- Si and Ge being gr-14 elements have 4 valence electron
- They form 4 covalent bonds with neighbour atoms
- Gr-15 elements have 5 valence electrons and occupy some lattice site
- 4 out of 5 electrons form covalent bonds
- The left out 1 electron is mobile and conducts electricity
- Conductivity is due to negatively charged electron
➡️ P-Type Semiconductor
- Intrinsic semiconductors when doped with gr-13 elements (B, Al, Ga), form p-type semiconductor
- Gr-13 elements have 3 valence electrons
- The 4th valence electron forms a hole
- This hole moves in the opposite direction of the electric field applied causing conductivity
- Conductivity is due to positively charged electron
? Applications of n-type and p-type semiconductors
- Diode is a combination of n and p-type semiconductor used for rectification of alternating current
- npn and pnp type of transistors are formed to detect or amplify radio or audio signals
- Used in solar cells and photodiode
? Magnetic Property
- The magnetic property of a material arises due to the revolving motion of electrons around the nucleus which can be considered as a small loop of magnet
- Each electron behaves as a tiny magnet.
- It happened due to the 1) orbital motion of electron around the nucleus 2) spin of electron in its own axis
- Magnitude of magnetic moment is measured in Bohr Magneton μB
- They are classified into 5 categories:
- Paramagnetic substances –

- Weakly attracted by the magnetic field
- Magnetized by magnetic field in the same direction
- They lose their magnetism in the absence of a magnetic field
- Arises due to the presence of unpaired electron
- Example – O2, Cu2+, Fe3+, Cr3+
2. Diamagnetic substances –
- Weakly repelled by the magnetic field
- Weakly magnetized in opposite direction in presence of a magnetic field
- Shown by substances that have paired electrons
- The pairing of electrons cancel their magnetic moments
- Example – H2O, NaCl, C6H6
3. Ferromagnetic substances –

- Strongly attracted by magnetic field
- Permanently magnetised
- In solid state, the metal ions are grouped together into small regions called domains
- Each domain acts as a tiny magnet
- The domains are randomly arranged in an unmagnetised piece of ferromagnetic substance
- Domains get aligned into the direction of magnetic field when exposed in it and a strong magnetic effect is produced
- Example – CrO2, Iron, Cobalt etc
4. Anti-Ferromagnetic substances –

- Domain is similar to ferromagnetic substances
- Their domains are oppositely oriented and cancel each others magnetic moment
- Example – MnO
5. Ferrimagnetic substances –
- Observed in particles where the domains are aligned in parallel and antiparallel directions in unequal number
- Weakly attracted by magnetic field
- Lose ferrimagnetism on heating and become paramagnetic
- Example – Fe3O4, MgFe2O4 etc
Frequently Asked Questions
Question 1: What is Isotropy?
Answer: Isotropy is the nature of a solid which means uniformity in all directions.
Their properties such as mechanical strength, refractive index, electrical conductivity remain the same in all directions.
Question 2: What is Solid?
Answer: A Solid State is one of the four fundamental states of matter which has a rigid structure and closely packed molecules.
Final Words
You have covered a detailed explanation of Solid State. We hope that above mentioned Solid State Notes will definitely help you in your exam.