Last updated on July 14th, 2024 at 05:40 pm
Acid Bases And Salt MCQ
Salts are group of ionic substances which are formed by the chemical reaction between an acid and base.
Below are some of the very important NCERT Acid Bases and Salts MCQ Class 10 Science Chapter 2 with answers. These Acid Bases and Salts MCQ have been prepared by expert teachers and subject experts based on the latest syllabus and pattern of CBSE Term 1 examination.
We have given these Acid Bases and Salts MCQ Class 10 Science Questions with Answers to help students understand the concept.
MCQ Questions for Class 10 Science are very important for the latest CBSE Term 1 and Term 2 pattern. These MCQs are very important for students who want to score high in CBSE Board, NEET and JEE exam.
We have put together these NCERT Acid Bases and Salts MCQ for Class 10 Science Chapter 2 with Answers for the practice on a regular basis to score high in exams. Refer to these MCQs Questions with Answers here along with a detailed explanation.

Acid Bases and Salts MCQ 1-25
1 . Identify the basic salt from the following salts
- Na2CO3
- NH4Cl
- NaNO3
- KCI
2. What is the nature of the new product which is formed by the action of water on quick lime?
- Neutral
- Acidic
- Amphoteric
- Basic
3. The pH range most conducive for life of fresh water plants and animals is
- 6.5-7.5
- 2.0-3.5
- 3.5-5.0
- 9.0-10.5
4. Phenolphthalein turns acids to solution.
- Colourful
- Colourless
- Pink
- Orange
5. Which of the following salts do not contain water of crystallisation?
- Blue vitriol
- Baking soda
- Washing soda
- Gypsum
6. Generally metals react with acids to give salt and hydrogen gas. Which of the following acids does not give hydrogen gas on reacting with metals (except Mn and Mg)?
- H2SO4
- HCI
- HNO3
- All of these
7. Which one of the following is acidic in nature?
- Gastric juice
- Sodium hydroxide solution
- Lime water
- Blood plasma
8. The chemical formula for plaster of Paris is
- CaSO4.2H2O
- CaSO4.H2O
- CaSO4.1/2H2O
- 2CaSO4.H2O
9. An aqueous solution ‘A’ turns phenolphthalein solution pink. On addition of an aqueous solution ‘B’ to ‘A’ and ‘B’
- A is strongly basic and B is a weak base.
- A is strongly acidic and B is a weak acid.
- A has pH greater than 7 and B has pH less than 7.
- A has pH less than 7 and B has pH greater than 7.
10. To protect tooth decay we are advised to brush our teeth regularly. The nature of the tooth paste commonly used is
- Acidic
- Neutral
- Basic
- Corrosive
11. Which one of these acid-base indicators is used by a visually impaired student?
- Litmus
- Turmeric
- Vanilla
- Phenolphthalein
12. When you add a few drops of acetic acid to a test-tube containing sodium bicarbonate powder, which one of the following is your observation?
- No reaction takes place
- A colourless gas with pungent smell is released with brisk effervescence
- A brown coloured gas is released with brisk effervescence
- Formation of bubbles of a colourless and odourless gas
13. A student puts a drop of reaction mixture of a saponification reaction first on a blue litmus paper and then on a red litmus paper. He may observe that
- There is no change in the blue litmus paper and the red litmus paper turns white.
- There is no change in the red litmus paper and the blue litmus paper turns red.
- There is no change in the blue litmus paper and the red litmus paper turns blue.
- No change in colour is observed in both the litmus papers
14. The pH value of a sample of hydrochloric acid is 2. pH value of this sample when diluted by adding water will be
- less than 2 but more than 0
- more than 2 but less than 7
- more than 7
- no change in pH
Click Below For All Class 10 Subjects Sample Papers 2024
15. While preparing soap a small quantity of common salt is generally added to the reaction mixture of vegetable oil and sodium hydroxide. Which one of the following may be the purpose of adding common salt?
- To reduce the basic nature of the soap
- To make the soap neutral
- To enhance the cleansing power of the soap
- To favour the precipitation of the soap
16. When a small amount of acid is added to water, the phenomena which occur are
(A) Dilution
(B) Neutralisation
(C) Formation of H3O+ ions
(D) Salt formation
The correct statements are:
- (A) and (C)
- (B) and (D)
- (A) and (B)
- (C) and (D)
17. Baking soda is a mixture of
- Sodium carbonate and acetic acid
- Sodium carbonate and tartaric acid
- Sodium hydrogen carbonate and tartaric acid
- Sodium hydrogen carbonate and acetic acid
- Unit- I: India And Contemporary World
- Chapter 1: The Rise of Nationalism in Europe
- Unit-II: Contemporary India-II
- Chapter 1: Resources and Development
- Chapter 3: Water Resources
- Chapter 4: Agriculture
- Unit III: Democratic Politics-II
- Chapter 1: Power Sharing
- Chapter 2: Federalism
- Unit IV: Economic
- Chapter 1: Development
- Chapter 2: Sectors of Indian Economy
18. Sodium hydrogen carbonate when added to acetic acid evolves a gas. Which of the following statements are true about the gas evolved?
(i) It turns lime water milky.
(ii) It extinguishes a burning splinter.
(iii) It dissolves in a solution of sodium hydroxide.
(iv) It has a pungent odour.
- (i) and (ii)
- (i), (ii) and (iii)
- (ii), (iii) and (iv)
- (i) and (iv)
19. If a few drops of a concentrated acid accidentally spills over the hand of a student, what should be done?
- Wash the hand with saline solution.
- Wash the hand immediately with plenty of water and apply a paste of sodium hydrogen carbonate.
- After washing with plenty of water apply a solution of sodium hydroxide on the hand.
- Neutralise the acid with a strong alkali.
20. Which one of the following types of medicines is used for treating indigestion?
- Antibiotic
- Analgesic
- Antacid
- Antiseptic
21. Which of the following is used for dissolution of gold?
- Hydrochloric acid
- Sulphuric acid
- Nitric acid
- Aqua regia
22. During the preparation of hydrogen chloride gas on a humid day, the gas is usually passed through the guard tube containing calcium chloride. The role of calcium chloride taken in the guard tube is to
- Absorb the evolved gas
- Moisten the gas
- Absorb moisture from the gas
- Absorb Cl– ions from the evolved gas
23. In an attempt to demonstrate electrical conductivity through an electrolyte, the following apparatus was set up. Which among the following statement(s) is (are) correct?

i. Bulbs will not glow because electrolyte is not acidic.
ii. Bulb will glow because NaOH is a strong base and furnishes ions for conduction.
iii. Bulb will not glow because the circuit is incomplete.
iv. Bulb will not glow because it depends upon the type of electrolytic solution.
- (i) and (iii)
- (ii) and (iv)
- (iv) only
- (ii) only
24. Match the chemical substances given in Column (A) with their appropriate application given in Column (B)
| Column (A) | Column (B) |
| A. Bleaching Powder | i. Preparation of glass |
| B. Baking Soda | ii. Production of H2 and CI2 |
| C. Washing Soda | iii. Decolourisation |
| D. Sodium Chloride | iv. Antacid |
- A-(ii) B-(i) c-(iv) D-(iii)
- A-(iii) B-(ii) c-(iv) D-(i)
- A-(iii) B-(iv) c-(i) D-(ii)
- A-(ii) B-(iv) c-(i) D-(iii)
25. 10 mL of a solution of NaOH is found to be completely neutralised by 8 mL of a given solution of HCI. If we take 20 mL of the same solution of NaOH, the amount of HCI solution (the same solution as before) required to neutralise it will be
- 4 mL
- 8mL
- 12mL
- 16 mL
MCQ Answers
1 . (1)
Na2CO3 is basic salt because it is formed by the reaction between strong base (sodium hydroxide, NaOH) and weak acid (carbonic acid, H2CO3).
2. (4)
When quick lime is added to water, it reacts to form slaked lime (Ca(OH)2). Slaked lime or calcium hydroxide is basic in nature because it dissociates into OH– ions when dissolved in aqueous solution.
3. (1)
A neutral pH is most conducive for life of fresh water plants and animals and pH range 6.5-7.5 is almost neutral.
4. (2)
Phenolphthalein is used as an indicator in acid-base titrations. For this application, it turns acids to colourless solutions and bases to pink solutions.
5. (2)
Baking soda is sodium bicarbonate (NaHCO3) in anhydrous form without any water of crystallisation.
6. (3)
Nitric acid (HNO3) does not give hydrogen gas on reacting with metals (except Mg and Mn) because it is a strong oxidising agent and oxidises the evolved H2 gas to water and itself gets reduced to the oxides of nitrogen. Whereas metals such as magnesium and manganese react with very dilute HNO3 to evolve 1-12 gas.
7. (1)
Gastric juice is acidic in nature because its pH ranges from 1.5-3.5.
8. (3)
The chemical formula of plaster of paris is CaSO4.1/2 1-H2O. The half a molecule of water in the formula means that two molecules of CaSO4 share one molecule of water so that the effective water of crystallisation for one Caso unit comes to half a molecule of water.
9. (3)
Phenolphthalein solution turns pink in basic solutions. Hence, solution A lies in the pH range of above 7. On adding solution B the pink colour disappears hence it has a pH less than 7.
10. (3)
When we consume acidic food or drinks, the pH of the mouth falls to 5.5 due to which tooth decay occurs. This lowering of pH within the mouth can lead to dental plaque. To prevent this, doctors advise to brush the teeth with the toothpaste which is basic in nature as it neutralises the acidic condition inside the mouth. Hence, the nature of the toothpaste should be basic.
11. (3)
Vanilla essence is used as an acid base indicator because of its smell which helps visually challenged people to realise the change in pH.
12. (4)
When a few drops of acetic acid is added to the test tube containing sodium bicarbonate powder, sodium acetate is formed along with the release of colourless and odourless CO2 gas which can be observed as the bubbles in the test tube.
13. (3)
Saponification is the alkaline hydrolysis of the fatty acid esters. Hence the solution is basic, bases with blue litmus gives no colour change. Whereas, on dipping red litmus paper in a mixture of saponification reaction it turns blue.
14. (2)
When we add water to the solution of hydrochloric acid (pH = 2) the pH will increase till 7 but it will be less than 7 as it contains more hydrogen ions (H+ ions) as compared to hydroxide ions (OH– ions). So, the pH on dilution will be more than 2 but less than 7.
15. (4)
A small quantity of common salt is generally added to the reaction mixture Of vegetable oil and sodium hydroxide to favour the precipitation of the soap because by adding salt to the suspension mixture, soap is precipitated as solid. This process is called salting out of soap.
16. (1)
When a small amount of acid is added to water, the phenomena which occur are dilution and formation of H3O+ ions. When water is added, a concentrated acid is turned into a dilute acid. It is process is called dilution of acid. A dilute acid is an aqueous solution in which acid undergoes ionisation and releases hydronium ions (H3O+).
17. (3)
Baking soda is a mixture of sodium hydrogen carbonate (NaHCO3) and mild edible tartaric acid.
18. (2)
Gas evolved ie carbon dioxide gas and it has the property to extinguish burning splinter. It can turn lime water milky and gets dissolved in a solution of sodium hydroxide to form sodium carbonate. It is an odourless gas.
19. (3)
If a few drops of a concentrated acid accidentally spills over the hand of a student then he should wash his hands immediately with plenty of water as it will dilute the acid and wash it out. Any remaining acid should be neutralised by applying the paste of sodium hydrogen carbonate.
20. (3)
Antacids such as sodium bicarbonate and magnesium hydroxide etc are used in treating indigestion. They are basic in nature and reacts with excess acid present in the stomach and neutralises it.
21. (4)
Aqua regia is a mixture of hydrochloric acid and nitric acid in the ratio 3:1 and it can dissolve noble metals such as gold, palladium, and platinum, which however, not soluble in either of the acids alone.
22. (3)
Calcium chloride is used as an absorbent in the guard tube during the preparation of hydrogen chloride gas on a humid day as it absorbs the moisture present in the air and gives out white fumes on reacting with moisture.
23. (2)
Bulb will glow because NaOH is a strong base that dissociates completely in water into Na+ ions and OH– ions which are responsible for electrical conductivity.
24. (3)
A-(iii) B-(iv) c-(i) D-(ii)
25. (4)
Given: 10 ml of NaOH requires 8 mL of HCI
Let X be the amount of HCI required to neutralise the 20 ml of NaOH
20 ml of NaOH requires = XHCI
X = 20×8/10
X = 16ml
Click Below For All Class 10 Subjects Sample Papers 2024
Assertion And Reasoning MCQs
- If both assertion and reason are true and reason is the correct explanation of assertion.
- If both assertion and reason are true, but reason is not the correct explanation of assertion.
- If the assertion is true, but the reason is false.
- If the assertion is false, but the reason is true.
1. Assertion: Solutions of compounds like alcohol and glucose do not show acidic character.
Reason: They do not show acidic character because they do not dissociate into ions.
2. Assertion: Dry HCI gas does not change the colour Of the dry litmus paper.
Reason: It is because dry HCI does not contain the OHions.
3. Assertion: Curd and sour substances should not be 47. stored in copper vessels.
Reason: Curd and other sour substances should not be kept in brass and copper vessels as they contain acids.
4. Assertion: Calcium starts floating when added in water.
Reason: Calcium starts floating because the bubbles of oxygen gas which are formed during the reaction stick to the surface Of the metal.
5. Assertion: Tap water conducts electricity but distilled water does not conduct electricity.
Reason: Tap water conducts electricity as it contains ions wheréas distilled water does not contain ions.
6. Assertion: Most of the metals do not give hydrogen while reacting with nitric acid.
Reason: Nitric acid is a weak oxidising agent.
7. Assertion: Plaster of Paris should be stored in a dry place.
Reason: To prevent its reaction with moisture.
8. Assertion: An aqueous solution of acid conducts electricity.
Reason: It is because in the solution it dissociates into ions.
9. Assertion: Ammonia is acidic in nature.
Reason: Ammonia dissolves in water and forms OH-.
10. Assertion: The aqueous solutions of glucose and alcohol do not show acidic character.
Reason: Aqueous solution of glucose and alcohol do not give H+ ions.
Assertion and Reasoning MCQ Answers
1 . (1)
Solutions of compounds like alcohol and glucose do not show acidic character because like acids such as HCI which dissociates into H+ and Cl– ion these solutions do not dissociate into ions.
2. (3)
Colour of the litmus paper is changed by the hydrogen ions. Dry HCI gas does not contain H+ ions. Acids give hydrogen ions only in the presence of ions. In case of dry litmus paper and dry HCI, water is not present. Therefore, the colour of the litmus paper does not change.
3. (1)
Curd and other sour substances should not be kept in brass and copper vessels as they contain acids. When these substances are kept in brass and copper vessels, the metal reacts with the acid to liberate hydrogen gas and harmful products are obtained due to which the food gets spoiled.
4. (3)
Hydrogen gas is released when calcium reacts with water. Calcium starts floating because the bubbles of hydrogen gas which are formed during the reaction stick to the surface of the metal. As a result, calcium starts floating on water.
5. (1)
Tap water conducts electricity as it contains ions whereas distilled water does not contain ions.
6. (3)
Nitric acid is a strong oxidising agent. Hydrogen produced during the reaction is oxidised to water and nitric acid itself gets reduced to nitrogen oxide.
7. (1)
Plaster of Paris is stored in a dry place because in the presence of water or moisture, it changes to a hard solid mass called gypsum.
8. (3)
Acids dissociate in aqueous solutions to form ions. These ions are responsible for conduction of electricity.
9. (4)
Ammonia is a weak base as it has one pair of electrons through which it can accept a proton.
10. (1)
The aqueous solution of glucose and alcohol do not dissociate to form the H+ ions in the solution thus they do not show the acidic character.
Case Study Based MCQs
1. Read the passage carefully and answer any four questions.
A compound C of sodium forms a white powder. It is a constituent of baking powder and is used in some antacids. When heated it gives a compound D which is anhydrous and absorbs water to become a hydrated salt. When this salt is kept in the open air, it loses water molecules in a process called efflorescence. When dissolved in water, it forms a strong base and a weak acid F.
(i) Which of the following is compound C?
- NaHCO3
- Na2CO3
- NaOH
- NaCl
(ii) Which of the following is compound D?
- NaHCO3
- Na2CO3
- Na2CO3 .10H2O
- NaCI
(iii) What is the nature of the solution formed by dissolving D in water?
- Acidic
- Alkaline
- Neutral
- Insoluble
(iv) Which among the following is compound F.
- CO2
- H2CO3
- NaOH
- H2O
(v) Sodium carbonate is a basic compound because it is a salt of a
- Strong acid and strong base
- Weak acid and weak base
- Strong acid and weak base
- Weak acid and strong base
2. Read the passage carefully and answer any four questions.

Acids show their property due to the presence of hydronium ions in their solution. They produce hydrogen ion (H+) in the presence of water.
Water weakens the bond between the ions and makes them soluble. This is the reason why acids and bases produce ions in aqueous solutions.
In non-polar solvents like benzene and toluene, dry HCI gas or solution does not undergo ionisation. The reason for this split is because the water molecules, being polar, pull the H+ and Cl– apart.
(i) A solution with pH = 8 will have the number of H+ ions:
- 108
- 10-8
- 1008
- log108
(ii) The correct order of acidic strength is:
- Water < Hydrochloric acid < Acetic acid
- Hydrochloric acid < Water < Acetic acid
- Acetic acid < Hydrochloric acid < Water
- Water < Acetic acid < Hydrochloric acid
(iii) Which of the following compound does not dissociate into H+ ions in an aqueous solution:
- C2H5OH
- H3PO4
- H2CO3
- CH3COOH
(iv) The pH value of four solutions are: A = 1 B= 9 C = 3 D=13
Choose the incorrect statement:
- A has a higher concentration of hydronium ions than solution C
- B has a higher concentration of hydronium ions than solution D
- A and B will turn red litmus solution into blue
- A is highly acidic while B is weakly basic
(v) Choose the incorrect statement
- An increase in pH value from 7 to 14 indicates an increase in hydrogen ions
- The strength of an acid or base is determined by a universal indicator
- pH < 7 indicates that the solution is acidic
- Higher the hydrogen ion concentration, lower the pH value
3. Read the following and answer any four questions.
Marble’s popularity began in ancient Rome and Greece, where white and off-white marble were used to construct a variety of structures, from hand-held sculptures to massive pillars and buildings.

(i) The substance not likely to contain CaCO3 is
- Dolomite
- A marble statue
- Calcined gypsum
- Sea shells
(ii) A student added 10g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
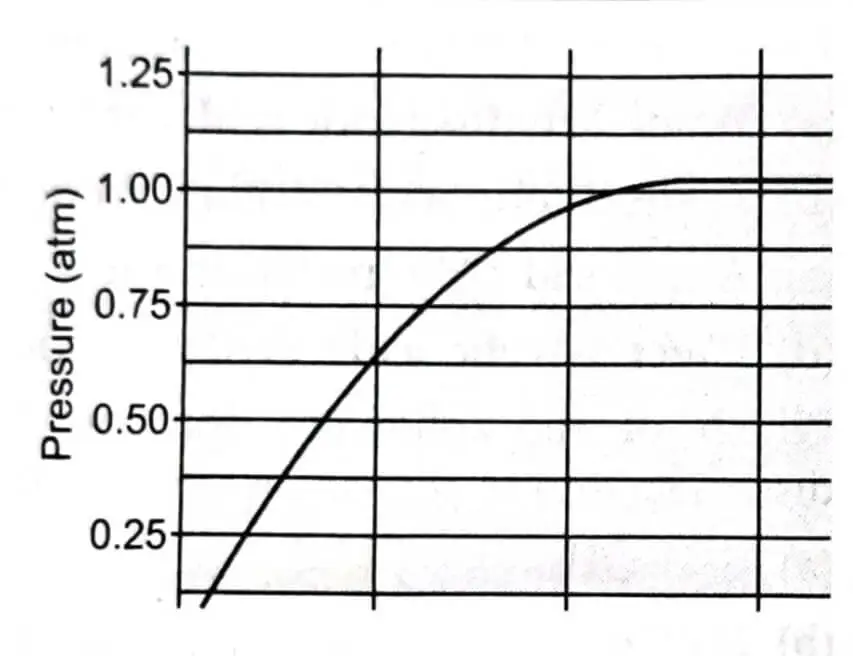
- 15-20 min
- 10-15 min
- 5-10 min
- 0-5 min
(iii) Gas A, obtained above is a reactant for a very important biochemical process which occurs in the presence of sunlight. Identify the name of the process:
- Respiration
- Photosynthesis
- Transpiration
- Photolysis
(iv) Marble statues are corroded or stained when they repeatedly come into contact with polluted rain water. Identify the main reason.

- decomposition of calcium carbonate to calcium oxide
- polluted water is basic in nature hence it reacts with calcium carbonate
- polluted water is acidic in nature hence it reacts with calcium carbonate
- calcium carbonate dissolves in water to give calcium hydroxide.
(v) Calcium oxide can be reduced to calcium, by heating the with sodium metal.Which compound would act as an oxidizing agent in the above process?
- Sodium
- Sodium oxide
- Calcium
- calcium oxide
4. Read the passage carefully and answer any four questions.
Common edible salt, NaCl (Sodium Chloride) obtained from seawater or from lakes contains many impurities such as sulphates of sodium and magnesium along with chlorides of calcium and magnesium.
The chlorides of these metals are particularly undesirable being deliquescent in nature. A saturated solution of salt in a minimum quantity of water is prepared and insoluble impurities are filtered off.
HCI gas is then passed through the saturated solution and the crystals of pure NaCl separate out. The soluble impurities remain in the solution. Pure crystals of NaCl are filtered, washed and dried.
(i) Choose the correct statement:
- Pure NaCl is hygroscopic in nature by itself
- Pure NaCl is soluble in alcohol
- NaCl shows hygroscopic properties only due to impurities
- NaCl is a brown crystalline solid
(ii) Nature of the aqueous solution of common salt is:
- Acidic
- Alkaline
- Basic
- Neutral
(iii) Which of the following compounds is alkaline in an aqueous medium?
- Na2CO3
- NaCl
- H2CO3
- CuSO4
(iv) Adding common salt to water will result in:
- Increase in pH of the water
- Decrease in pH of the water
- No change in pH of the water
- Flames in water
(v) Examine a few statements regarding NaCl
(I) It is prepared by Chlor-alkali process
(II) It is a white crystalline substance
(III) It also exists in the form of rocks called rock salt
(IV) It is a neutral salt with a pH value = 7
- II and Ill only
- III and IV only
- I and IV only
- Il, Ill and IV only
5. Read the following and answer any four questions.
Reshma broke her leg in an accident. She went to see Dr. Sant Prakash. On examination, Dr. Prakash mixed the white power in water and applied to her leg along with the cotton and gauze. After a while, it turned into white, solid, hard mass. He said that it would support her fractured bone in the right position.
(i) After treatment, the doctor repacked the white powder back into a moisture proof, airtight container. Why?
- The fungus growth will occur in open.
- powder would react to moisture and turn into solid mass.
- powder will react to sunlight and turn into solid mass.
- To prevent the stealing of the powder as it is very expensive.
(ii) What is ‘white, solid hard mass’ called as?
- Talcum powder
- Plaster of Paris
- Paris of Plaster
- Copper sulphate
(iii) The reaction involved in the formation of white mass is:
- Combustion
- Mineralisation
- Oxidation
- Crystallisation
(iv) The graph shows the porosity and expansion of plaster with respect to water content. At what temperature, the reaction would occur?
- 373K
- 673K
- 273K
- 573K
(v) Study the following reaction and choose the correct option:
CaSO4 . ½ H2O + 3/2 H2O → CaSO4.2H2O
- Reactant is calcium hemihydrate, the product is gypsum.
- Reactant is gypsum, product is calcium hemihydrate.
- Reactant is gypsum, product is calcium sulphate hemihydrate.
- Reactant is calcium sulphate hemihydrate, the product is gypsum.
6. Read the passage carefully and answer any four questions.
POP or Plaster of Paris is a calcium sulphate hemihydrate. Hemihydrate contains half a molecule of water of crystallization. It is represented by the formula CaSO4.1/2H2O.
This structure of half water molecules means that one water molecule is shared by two formula units of CaSO4. The name Plaster of Paris was given to this compound because, for the first time, it was made from gypsum which was mainly found in Paris.
(i) The difference of water molecules in gypsum and Plaster of Paris is
- 5/2
- 2
- 1/2
- 3/2
(ii) Plaster of Paris is known for its hardening. It is due to
- releasing CO2
- converting into CaCO3
- combining with water
- losing out water
(iii) Choose the incorrect statement
- POP is used to ornate designs on walls and ceilings
- On heating gypsum above 373 K, CaSO4 is obtained
- Dead burnt plaster is CaSO4.2H2O
- The setting of plaster is due to its hydration into gypsum
(iv) Choose the incorrect statement related to gypsum:
- It is slightly soluble in water
- It is also known aq alabaster
- On heating gypsum at 373 K. It loges water molecules and becomes calcium sulphate hemihydrate
- The chemical formula of gypsum is CaSO4.½H2O
(v) We can obtain Plaster of Pari a by
- Adding water to calcium sulphate
- Adding sulphuric acid to calcium hydroxide
- Heating gypsum to a very high temperature
- Heating gypsum to 1000C
7. Answer the following questions on the basis of your understanding of the passage.
| S. No. | Substances | pH value |
| 1 | Lemon juice | 2 |
| 2 | Wine | 4 |
| 3 | Detergent | 10 |
| 4 | Beach | 12.5 |
(i) What is the nature of bleach?
(ii) What is the colour of litmus for lemon juice?
(iii) What is the pH value of neutral solutions?
(iv) Name the substances that would turn red litmus blue.
8. A white shirt has a yellow stain of curry. When soap is rubbed on this shirt during washing, the yellow stain turns reddish-brown. On rinsing the shirt with plenty of water, the reddish-brown stain turns yellow again.
(i) Name the natural indicator present in curry stains.
(ii) Explain the changes in colour of this indicator which take place during washing and rinsing the shirt.
(iii) What is the nature of soap (acidic/basic) as shown by the indicator present in curry stains?
Click Below For All Class 10 Subjects Sample Papers 2024
9. Solution A turns the universal indicator blue to purple whereas solution B turns the universal indicator orange to red.
(i) What will be the action of solution A on litmus?
(ii) What will be the action of solution B on litmus?
(iii) Name any two substances which can give solutions like A.
(iv) Name any two substances which can give solutions like B.
(v) What sort of reaction takes place when solution A reacts with solution B?
10. Based on the table given below answer the following questions
| S. No. | Column I | Column II |
| 1 | Tartaric acid | Calcium hydroxide |
| 2 | Lactic acid | Sodium hydroxide |
| 3 | Formic acid | Ammonia |
| 4 | Citric acid | Magnesium |
(i) What does column I represent?
(ii) What does column Il represent?
(iii) Name one source of citric acid.
(iv) What is released by an ant’s sting?
Case Based MCQs Answers
1.
(i) (1) NaHCO3
(ii) (2) Na2CO3
(iii) (2) Alkaline
(iv) (2) H2CO3
(v) (4) Weak acid and strong base
2.
(i) (2) 10-8
(ii) (4) Water < Acetic acid < Hydrochloric acid
(iii) (1) C2H5OH
(iv) (1) A has a higher concentration of hydronium ions than solution C
(v) (1) An increase in pH value from 7 to 14 indicates an increase in hydrogen ions
3.
(i) (4) Sea shells
(ii) (4) 0.5 mm
(iii) (2) Photosynthesis
(iv) (3) polluted water is acidic in nature hence it reacts with calcium carbonate
(v) (4) calcium oxide
4.
(i) (3) NaCl shows hygroscopic properties only due to impurities
(ii) (4) Neutral
(iii) (1) Na2CO3
(iv) (3) No change in pH of the water
(v) (4) II, III and IV only
5.
(i) (2) powder would react to moisture and turn into solid mass
(ii) (2) Plaster of Paris
(iii) (4) Crystallisation
(iv) (1) 373K
(v) (4) Reactant is calcium sulphate hemihydrate product is gypsum.
6.
(i) (4) 3/2
(ii) (3) Combining with water
(iii) (3) Dead burnt plaster is CaSO4.2H2O
(iv) (4) The chemical formula of gypsum is CaSO4.1/2H2O
(v) (4) Heating gypsum to 100°C
7.
(i) Bleach is basic in nature.
(ii) Lemon juice turns blue litmus red.
(iii) The pH value for neutral solutions is 7.
(iv) Detergent and bleach would turn red litmus blue.
8.
(i) Turmeric.
(ii) The yellow stain of curry turns reddish-brown when soap is scrubbed on it because of the fact that soap solution is basic in nature which changes the colour of turmeric in the curry stain to red-brown. This stain turns yellow again when the cloth is rinsed with water because then the basic soap gets removed with water.
(iii) Basic.
9.
(i) Solution A turns universal indicator blue to purple so it is basic in nature and will turn litmus blue.
(ii) Solution B turns the universal indicator orange to red so it is acidic in nature and will turn litmus red.
(iii) Milk of magnesia and sodium hydroxide solution are bases like solution A.
(iv) Lemon juice and hydrochloric acid are acids like solution B.
(v) Neutralisation reaction.
10.
(i) Column I represents acids.
(ii) Column Il represents bases.
(iii) Oranges are a good source of citric acid.
(iv) Formic acid is released by ant’s sting.
Click Below For All Class 10 Subjects Sample Papers 2024
- Unit- I: India And Contemporary World
- Chapter 1: The Rise of Nationalism in Europe
- Unit-II: Contemporary India-II
- Chapter 1: Resources and Development
- Chapter 3: Water Resources
- Chapter 4: Agriculture
- Unit III: Democratic Politics-II
- Chapter 1: Power Sharing
- Chapter 2: Federalism
- Unit IV: Economic
- Chapter 1: Development
- Chapter 2: Sectors of Indian Economy
Final Words
From the above article, you have practiced Acid Bases and Salts MCQ of Class 10 Science Chapter 2. We hope that the above mentioned latest Chemical Reactions and Equations MCQ for Term 1 of Chapter 1 will surely help you in your exam.
If you have any doubts or queries regarding the Acid Bases and Salts MCQ with Answers of CBSE Class 12 Physics, feel free to reach us and we will get back to you as early as possible.
Click Below To Learn Physical Education Class 12 Term-1 Syllabus Chapters MCQs
- Chapter 1: Planning in sports MCQ
- Chapter 2: Sports And Nutrition MCQ
- Chapter 5: Children and Women in Sports MCQ
- Chapter 6: Test and Measurement in Sports MCQ
- Chapter 8: Biomechanics and Sports MCQ